Cholera infections caused by Vibrio cholerae bacteria can be life-threatening and the trigger is the cholera toxin produced by the bacteria. It binds to the surface of intestinal cells—more precisely, to certain “sugar lipids” (GM1 gangliosides, GM1) on the cell surfaces. This bond is one of the strongest known interactions between a protein—the toxin—and the sugar part of GM1. It enables the cholera toxin to penetrate the intestinal cells, which causes the very rapid loss of fluid.
In an interdisciplinary approach, a team from the University of Münster, ETH Zürich and Leibniz Universität Hannover has now analyzed a key component of the GM1 cholera toxin complex for the first time using a fluorinated GM1 analog. The findings on the molecular mechanisms of the strong interaction help to better understand the disease and could enable the development of novel drugs.
The work was published in the journal ACS Central Science.
As one of the most abundant biomolecule classes on the plant, carbohydrates are essential in all areas of biology and medicine. From determining blood groups to regulating the immune system and supplying cells with energy—the complexity of these sugar molecules offers great potential for the development of next-generation drugs. However, their interaction with the target proteins is often too weak to be utilized for therapeutic purposes.
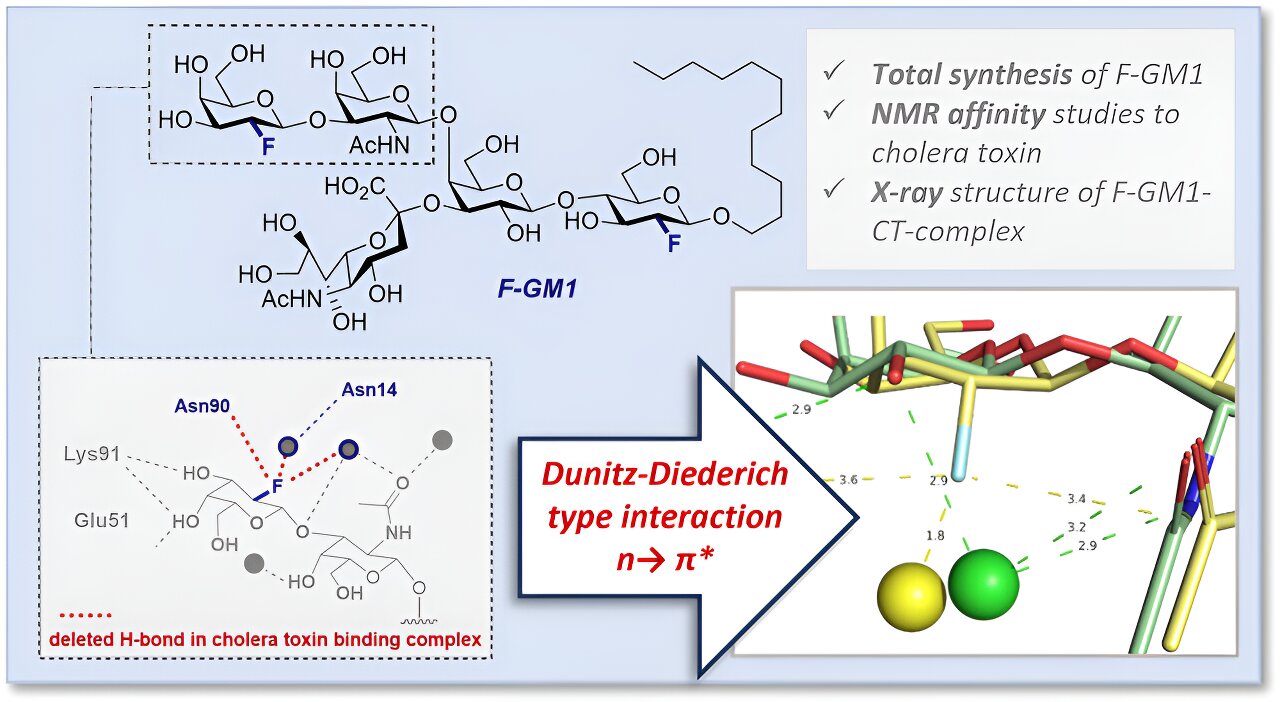
One exception is the previously mentioned strong interaction between the GM1 ganglioside and cholera toxin, which has been intensively investigated for decades. In order to be able to characterize this interaction more precisely at the molecular level, the research team produced a fluorinated GM1 analog (F-GM1) via a complex chemical synthesis. Fluorine served as a tool to identify the molecular interactions of this so-called glycomimetic.
There are several advantages of fluorine as a substitute for a hydroxyl group (OH): increased stability of the molecule against enzymatic degradation, control of the spatial arrangement of the atoms (stereoselectivity) during chemical glycosylation and the possibility of investigating the interaction of cholera toxin with F-GM1 in solution, i.e. in a state similar to that in the body, by nuclear magnetic resonance spectroscopy (NMR spectroscopy). The affinity—i.e. the desire to form a bond—of the analog was close to the affinity and binding strength of natural GM1.
The researchers also used protein crystallography to investigate the interactions and spatial arrangement of the atoms of F-GM1 within the binding pocket in more detail in order to explain the slightly lower affinity for choleratoxin compared to the natural sugar. In doing so, they demonstrated an additional interaction within the molecular complex caused by the fluorine as well as a changed arrangement of some functional groups in the binding pocket.
The study highlights the potential of using fluorinated gangliosides in biomedical research, for example to investigate molecular signaling cascades involving sugar chains (glycans), to identify possible new active substances or for vaccine development.
More information:
Christina Jordan et al, Probing the Origin of Affinity in the GM1-Cholera Toxin Complex through Site-Selective Editing with Fluorine, ACS Central Science (2024). DOI: 10.1021/acscentsci.4c00622
Citation:
Interdisciplinary approach provides new insights into molecular mechanisms of cholera infection (2024, July 15)
retrieved 15 July 2024
from https://phys.org/news/2024-07-interdisciplinary-approach-insights-molecular-mechanisms.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.












/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer_public/d1/82/d18228f6-d319-4525-bb18-78b829f0791f/mammalevolution_web.jpg)






Discussion about this post