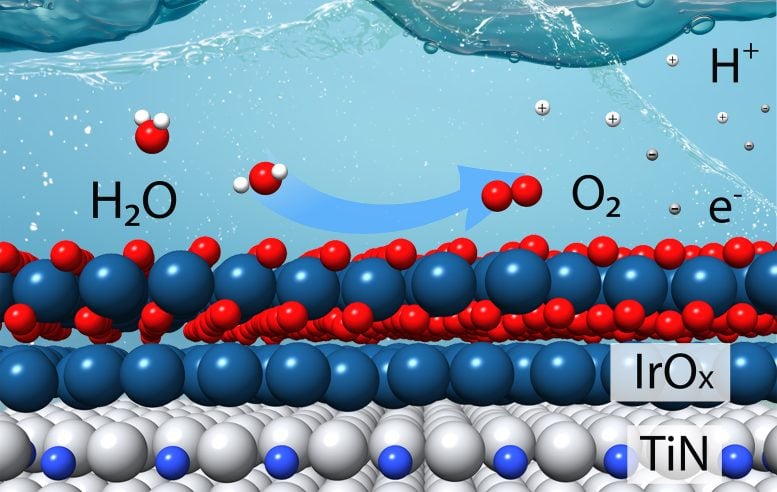
This schematic shows how a catalyst composed of a few layers of iridium oxide (IrOx) over a support made of titanium nitride (TiN) can efficiently produce oxygen (O2), hydrogen ions (H+), and electrons (e–) from water molecules (H2O) in an acidic electrolyte. This “oxygen evolution reaction” is the more challenging of two reactions needed to split water to produce hydrogen gas (H2). Credit: Tianyou Mou/Brookhaven National Laboratory
A successful demonstration could enhance the production of hydrogen from water.
Hydrogen (H2) holds great potential as a fuel to reduce greenhouse gases, particularly when produced by using renewable energy to split water molecules (H2O). However, despite the apparent simplicity of breaking water into hydrogen and oxygen, the underlying chemistry is quite complex.
Two separate simultaneous electrochemical reactions each require catalysts, chemical “deal makers” that help break and remake chemical bonds. Now, scientists at the U.S. Department of Energy’s (DOE) Brookhaven National Laboratory and Columbia University say they’ve developed a new efficient catalyst for the more challenging part: the oxygen evolution reaction.
As described in a paper just published in the Journal of the American Chemical Society, the catalyst was designed “from the bottom up” based on theoretical calculations seeking to minimize the amount of iridium, an expensive metal used as a catalytic material, and to maximize the catalyst’s stability in acidic conditions. When the team created models of the catalyst and tested them in the lab, the results validated the predictions. Then, the scientists made a powder form of the catalyst, like those used in industrial applications, and showed it can efficiently produce hydrogen in a water-splitting electrolyzer.
“In this real-world test, our catalyst is about four times better than the state-of-the-art commercially available iridium catalyst,” said Jingguang Chen, a chemical engineer at DOI: 10.1021/jacs.4c02936
This work was funded by the DOE Office of Science. CFN, NSLS-II, and NERSC all operate as DOE Office of Science user facilities.
Brookhaven National Laboratory is supported by the Office of Science of the U.S. Department of Energy


