Peptides are biomolecules shaped when two or extra amino acids that carry out key capabilities within the human organism, corresponding to hormones, neurotransmitters, painkillers and antibiotics, bind collectively. For that reason, they’re much studied and utilized by the pharmaceutical trade, for instance.
A research performed by scientists within the Division of Biophysics on the Federal College of São Paulo’s Medical Faculty (EPM-UNIFESP) in Brazil recognized important modifications within the physicochemical properties of peptides throughout a spontaneous technique of chemical change referred to as pyroglutamination.
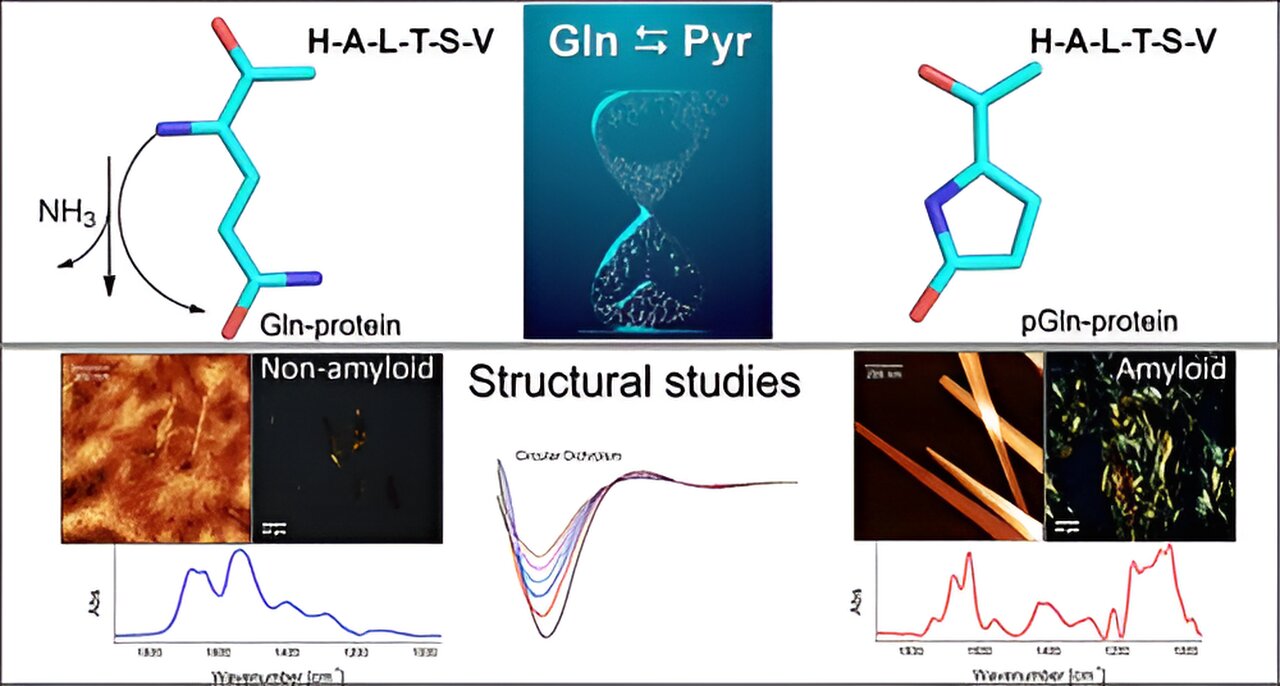
Pyroglutamination is a modification ensuing from spontaneous conversion of glutamine to pyroglutamic acid, with a major affect on the bodily and chemical properties of peptides. It’s a well-known however incessantly neglected a part of peptide synthesis, and infrequently explored in proteomics.
The researchers who performed the research stress that it may possibly happen quickly and accelerates as temperature rises, underscoring the necessity for warning throughout laboratory experiments to forestall glutamine cyclization. It’s particularly vital in situations that mimic physiological environments the place temperatures are within the vary of 37° C, the conventional temperature of a wholesome human organism.
The invention has implications for laboratory analysis and opens up new prospects for the research of neurodegenerative illnesses corresponding to Alzheimer’s and Parkinson’s, since after chemical modification the molecule acquires an amyloidal construction, which favors aggregation of molecules, forming plaques like these believed to trigger the illnesses in query.
An article on the research is printed in Biochemistry.
The group performed in vitro experiments to analyze the mechanism whereby the amino acid glutamine (Gln) turns into pyroglutamic acid (Pyr) within the presence of a peptide or protein sequence on the N-terminal extremity. This course of happens by way of deamidation, a response that eliminates ammonia (NH3). Pyr (additionally referred to as pyroglutamate) is a cyclic amino acid shaped because of dehydration of glutamate. All proteins encompass a number of amino acids joined collectively by peptide bonds, with variations within the quantity and sequence of amino acids.
“The outcome can function a mannequin for a lot of researchers who work with peptides. We arrived at two key findings. We returned to an previous matter, which is how glutamine breaks down into pyroglutamic acid, however we launched a warning in regards to the significance of analyzing the sequence. The second level was that after conversion of the peptide, its traits change and it tends to stay to membranes.”
“The presence of pyroglutamic acid favors the formation of amyloidogenic aggregates, just like the conglomerates usually present in circumstances of neurodegenerative illness. These amyloid plaques are shaped within the mind and interrupt the move of neurons,” stated Clovis Ryuichi Nakaie, final creator of the article.
Levels of the analysis
The mannequin peptide sequence (QHALTSV-NH2) used within the research originated within the Ph.D. analysis of Mariana Machado Leiva Ferreira, first creator of the article, whereas she was on the lookout for a synthesis of some two dozen peptides current within the sequences of 5 G-protein coupled receptors (GPCRs) that diverse in dimension as much as about 20 amino acids. GPCRs seize a wide selection of extracellular indicators (starting from photons to ions, proteins, neurotransmitters and hormones) and activate signaling pathways inside cells.
One of many peptides synthesized by Ferreira stood out for its low yield and was the one one with glutamine on the amine extremity. “After the primary try at synthesis with a really low yield, we diverse a number of parameters to extend manufacturing of the peptide, together with modifications to the artificial half and to the purification course of, however sadly it all the time partially degraded,” she stated.
When the group examined options incessantly utilized in proteomic experiments, they discovered that glutamine conversion to pyroglutamic acid occurred in all of them as a perform of time, in accordance with typical first-order kinetics, the place the speed of conversion was proportional to the time taken by the response. They then determined to not agitate the answer in order that the dialog fee might be inferred. For instance, they estimated that after 5 hours at the least 10% of the glutamine in all probability transformed into pyroglutamic acid.
A minor structural change triggered when the native peptide was pyroglutaminated on the N-terminal extremity was adequate to alter the molecule’s physicochemical conduct.
“As a result of it is cyclical and has one much less optimistic cost, the peptide Pyr must be extra hydrophobic than the native molecule, and we subsequently anticipated the analog to work together with membrane-mimetic techniques. What we did not foresee was that the analog would entail the formation of amyloid buildings like these seen in neurodegenerative illnesses. We did not research any of those, however our outcomes level in that path,” Emerson Rodrigo da Silva, penultimate creator of the article, informed Agência FAPESP. Silva and Nakaie are the corresponding authors.
Nakaie burdened the significance of post-translational modifications within the organism involving the polypeptide chain. They play a task within the useful range of proteins and allow adaptation of a sequence of amino acids encoded by a gene to carry out varied regulatory capabilities.
“On this context, time as an element will all the time correlate with the incidence of modifications, no matter their pace or their location in our organism. This remembers the concept of the organic clock which explains why we instructed placing an hourglass on the quilt of the journal to represent the spontaneous conversion of Gln into Pyr,” Nakaie stated.
He has been a professor at EPM-UNIFESP for 45 years and burdened the groundbreaking work executed by the group within the Division of Biophysics. Specifically, he famous, they launched the synthesis and biochemistry of peptides and amino acid derivatives to Brazil.
“Our findings will undoubtedly pave the best way for additional research. After finishing the work of which Mariana Ferreira’s Ph.D. analysis was half, we additionally wish to go on with this analysis line,” he stated.
Extra info:
Mariana M. L. Ferreira et al, Pyroglutamination-Induced Adjustments within the Physicochemical Options of a CXCR4 Chemokine Peptide: Kinetic and Structural Evaluation, Biochemistry (2023). DOI: 10.1021/acs.biochem.3c00124
Quotation:
Chemical course of makes peptide purchase construction just like amyloid plaques present in neurodegenerative illnesses (2023, November 1)
retrieved 1 November 2023
from https://phys.org/information/2023-11-chemical-peptide-similar-amyloid-plaques.html
This doc is topic to copyright. Aside from any honest dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is offered for info functions solely.