By
A breakthrough in autism research has been achieved with the CHOOSE system, developed by IMBA and ETH Zurich researchers. This technology enables detailed study of genetic mutations in human brain organoids, offering profound insights into autism’s underlying mechanisms and paving the way for broader disease research applications.
The CHOOSE system, an innovative approach combining brain organoids and genetics, transforms autism research by allowing detailed analysis of mutations and their effects on brain development.
Does the human brain have an Achilles heel that ultimately leads to Autism? With a revolutionizing novel system that combines brain organoid technology and intricate genetics, researchers can now comprehensively test the effect of multiple mutations in parallel and at a single-cell level within human brain organoids.
This technology, developed by researchers from the Knoblich group at the Institute of Molecular Biotechnology (IMBA) of the Austrian Academy of Sciences and the Treutlein group at ETH Zurich, permits the identification of vulnerable cell types and gene regulatory networks that underlie autism spectrum disorders. This innovative method offers unparalleled insight into one of the most complex disorders that challenge the human brain with implications that bring autism clinical research much-needed hope.
Human Brain Development and ASD Risks
Compared to other animal 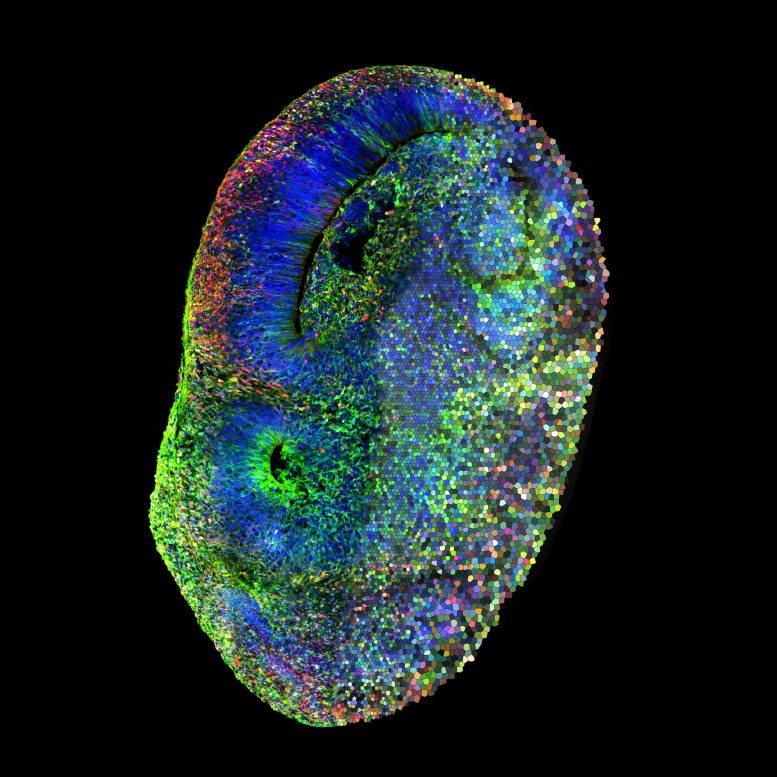
Left half: Confocal image of a CHOOSE (CRISPR-human organoids-scRNA-seq) human brain organoid mosaic system showing cells carrying a mutation in red. Right half: a mosaic depiction of different colors representing single cells, each carrying a mutation in one high-confidence autism gene. Credit: ©Knoblich Lab / IMBA-IMP Graphics
Innovative CHOOSE System for Genetic Screening
To help crack this black box open, researchers from Jürgen Knoblich’s and Barbara Treutlein’s research groups at IMBA and ETH Zurich developed a technique to screen a complete set of key transcriptional regulator genes linked to autism. This development is especially impactful since the genes of interest can be examined simultaneously within a single mosaic organoid, marking the beginning of an era of intricate, efficient, and expedient genetic screening in human tissue.
In the newly developed system, called “CHOOSE” (CRISPR-human organoids-scRNA-seq), each cell in the organoid carries at most one mutation in a specific ASD gene. The researchers could trace each mutation’s effect at a single-cell level and map each cell’s developmental trajectory.
“With this high-throughput methodology, we can systematically inactivate a list of disease-causing genes. As the organoids carrying these mutations grow, we analyze the effect of each mutation on the development of each cell type,” says the study’s first and co-corresponding author Chong Li, a postdoctoral fellow in the Knoblich group.
A High-Throughput Systematic Approach
With the CHOOSE system, the IMBA and ETH Zurich teams advance research on disease-causing genes by a whole leap, providing researchers with access to a versatile and high-throughput method that can be applied to any disease and in any human model system. Importantly, CHOOSE considerably speeds up the analysis in comparison to traditional genetic loss-of-function approaches.
“We can see the consequence of every mutation in one experiment, thus shortening the analysis time drastically when compared to traditional methods, using an approach that for decades was only possible in organisms like the fruit fly”, explains Knoblich. “Additionally, we can still benefit from a hundred years of scientific literature about disease-causing genes.”
Mutating several genes in parallel and tracking their effects generates an enormous amount of data. To analyze this complex dataset, co-corresponding author Barbara Treutlein and her team at ETH Zurich used quantitative bioinformatics and DOI: 10.1038/s41586-023-06473-y
IMBA Scientific Director Jürgen Knoblich is also a Professor in Synthetic Biology at the Medical University of Vienna.
Funding: Simons Foundation Autism Research Initiative, Austrian Federal Ministry of Education, Science and Research, City of Vienna, Austrian Science Fund, European Research Council, Chan Zuckerberg Initiative, Silicon Valley Community Foundation, Swiss National Science Foundation, National Centre of Competence in Research Molecular Systems Engineering, European Molecular Biology Organization


