New research revealed that cell nuclei, not just cell surfaces, control the stiffness and organization of eye and brain tissues during development, indicating a significant role for the nucleus in shaping embryonic tissue structure and potentially influencing related diseases.
Biophysicist Otger Campàs and his research team, conducting research at both UC Santa Barbara and the Physics of Life Excellence Cluster at TU Dresden, have discovered that cell nuclei play a crucial role in shaping the architecture and mechanics of eye and brain tissues during embryonic development. This finding reveals a new function for the cell nucleus in tissue organization, extending beyond its traditional role in genetic regulation.
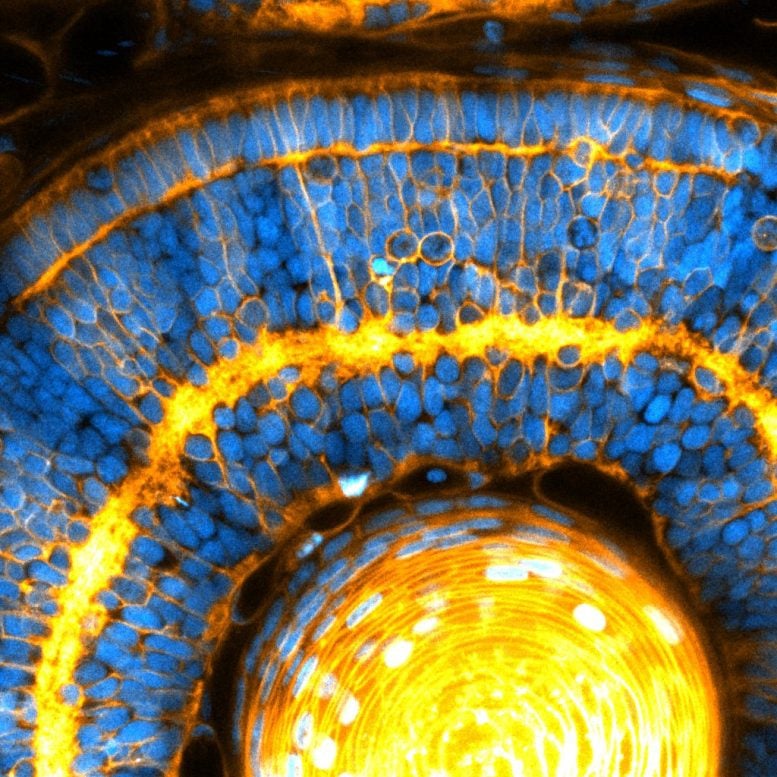
“We were measuring tissue stiffness in the zebrafish retina, and realized that it depended on the packing of nuclei. This was totally unexpected because tissue mechanics is believed to depend on cell surface interactions, but not organelles inside cells,” said Campàs, now professor and the chair of tissue dynamics at the Cluster of Excellence Physics of Life at TU Dresden, where he also serves as managing director. This research, published in the journal Nature Materials, represents an unexplored avenue to understand how cells orchestrate embryonic development.
The Hidden Architect
Inside each cell, individual structures known as organelles perform key functions, but how these organelles contribute to the formation of tissues and organs is unknown. Like factories or roads in cities, myriad organelles perform tasks inside cells for them to properly function. Because they are confined within cells, organelles were not believed to play a direct role in building organs during embryogenesis. Until now.
The cell’s nucleus is an organelle known for processing information in cells, with genes turning on and off depending on the signals received. However, the nucleus is also the largest and stiffest organelle in cells, and could affect the physical structure of the tissue in addition to processing information. Fascinated with how the nucleus could play a role in tissue formation, Campàs decided to study the role of nuclei in the formation of organs.
Previous pioneering work by his group had found that cell collectives acted like a foam during development that could be either jammed to “freeze” the tissue architecture and set its shape, or ‘melted’ to allow tissues to flow and shape them.
“By extending the Active Foam Model, we identified a new mode of solid-to-fluid transition, governed by the relative nucleus and cell sizes,” said co-lead author Sangwoo Kim.
When the authors investigated the size of the nucleus compared to that of cells in eye and brain tissues in both experimental and theoretical settings, they found that if the nucleus was taking up much of the cell space, then the tissue stiffness was directly controlled by the nucleus. Moreover, they also found that when the nuclei packed so strongly, they ordered the cells into nearly crystalline arrays.
“When the nuclei start to interact mechanically, both tissue mechanics and cellular ordering are not dictated by the cell surface, but rather controlled by the nucleus itself,” Campàs said. “This is an organelle determining the stiffness of the entire tissue.” Their study challenges the status quo, revealing a new role for nuclei in the control of tissue organization and mechanics.
To explore how the size of the cell’s nucleus affects organ formation, the researchers employed zebrafish. These DOI: 10.1038/s41563-024-01972-3


