
Scientists have developed methods to control the precise shapes and “handedness” of peptoids, protein-like molecules inspired by natural molecular structures, which are vital for applications like drug delivery and disease therapy. Their research highlights their innovative techniques in creating functional peptoid-based materials for various technological and medical uses. Credit: Illustration by Nathan Johnson | Pacific Northwest National Laboratory
Researchers have learned to control the “handedness” of peptoids, a crucial factor in precision drug delivery and diagnostics.
Nature is filled with extraordinarily precise molecular shapes that fit together like a hand in glove. Proteins, for example, can assemble into a wide variety of well-defined shapes that grant them their function.
“Depending on their shape, proteins can fit together with other proteins to perform functions or malfunction by clumping together, as observed in 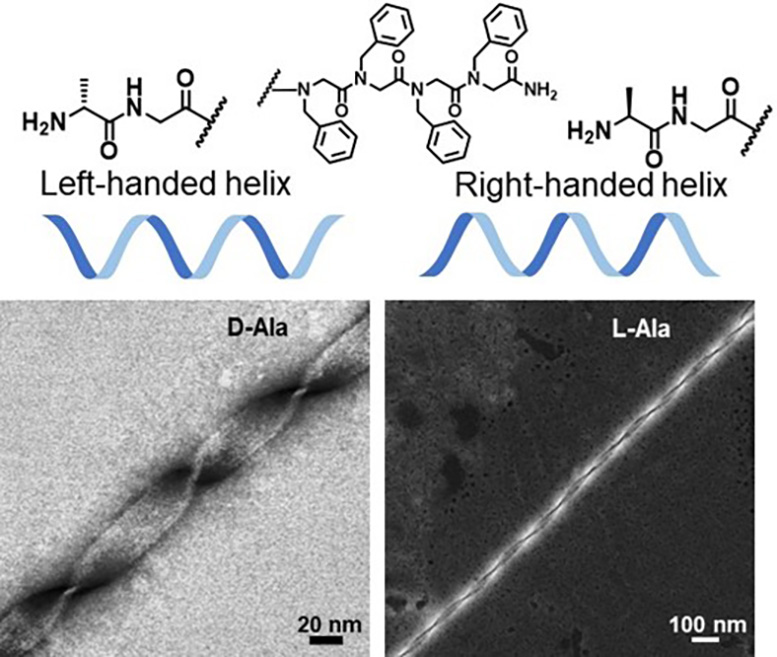
Chun-Long Chen and his team developed a way to control the shape of the peptoid helix. Credit: Chun-Long Chen | Pacific Northwest National Laboratory
For this experiment, Chen and his team chose to pursue corkscrew-like helical structures because of their biological importance. In fact, most proteins contain these basic helical structures.
Previous peptoid synthesis methods would yield a mix of left- and right-handed helices. In nature, proteins need to be in a specific conformation to perform their functions—most being left-handed.
“Other groups before us were able to synthesize peptoid nanohelices, but precisely controlling their shapes and handedness remained a challenge,” said Chen. “Being able to control their shapes would not only open the door for designing future materials, it would also provide insights into biological processes involving these structures.”
Using a combination of experimental and computational techniques, Chen and his team discovered a way to control the handedness of a peptoid helix. Similar to proteins, peptoids are created from amino DOI: 10.1038/s41467-024-46839-y
“Hierarchical Self-Assembly of Multidimensional Functional Materials from Sequence-Defined Peptoids” by Li Shao, Dehong Hu, Shao-Liang Zheng, Thi Kim Hoang Trinh, Wenhao Zhou, Haoyu Wang, Yanxu Zong, Changning Li and Chun-Long Chen, 24 April 2024, Angewandte Chemie International Edition.
DOI: 10.1002/anie.202403263
Both studies were primarily supported by the Department of Energy, Office of Science, Basic Energy Sciences program as part of the Energy Frontier Research Centers program: CSSAS – The Center for the Science of Synthesis Across Scales.


